GALEAS™ Bladder
Ultra-Sensitive, Urine-Based Bladder Cancer Test
Revolutionizing Bladder Cancer Diagnosis, Detection, and Screening
GALEAS™ Bladder is a cutting-edge, non-invasive bladder cancer test developed through a collaboration between researchers at the University of Birmingham in the UK and Nonacus. Utilizing advanced next-generation sequencing (NGS) technology, this test detects key somatic mutations associated with all grades and stages of bladder cancer. With its ultra-sensitive NGS technology, GALEAS™ Bladder accurately identifies tumor-derived gDNA in urine, offering a reliable and painless alternative to traditional diagnostic methods.
Why Choose GALEAS™ Bladder?
- Ultra-Sensitive Detection
-
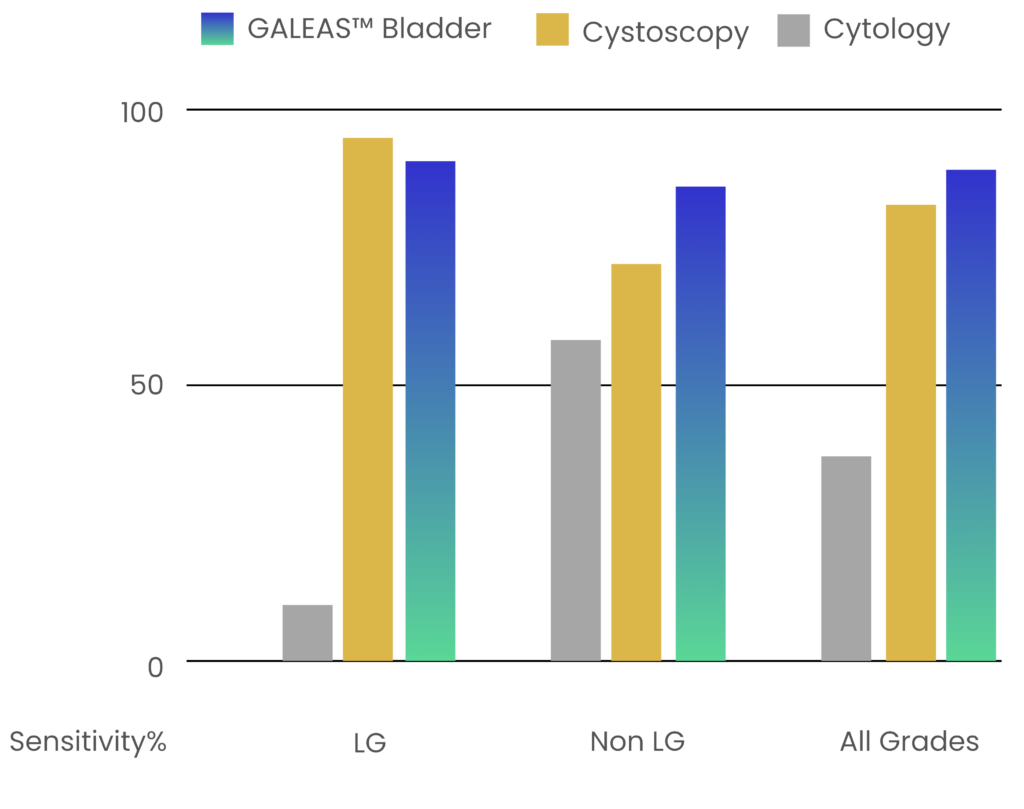
Identifies somatic mutations across 23 genes present in over 96% of bladder cancer cases
- Non-Invasive
-
Simple urine-based test eliminates unnecessary invasive procedures like cystoscopy
- Utilized Across Study Cohorts
-
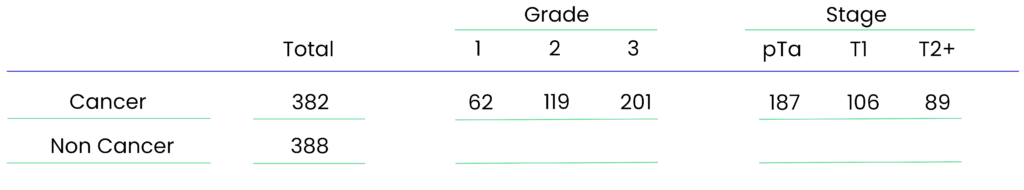
Tested on 710 urine samples across three UK clinical cohorts, ensuring high accuracy and reliability
- Comprehensive Reporting
-
Provides detailed somatic variant analysis and positive/negative results
Watch the GALEAS™ Bladder Video Introduction
Performance Data
GALEAS™ Bladder delivers exceptional performance across all stages and grades of bladder cancer:
| Sensitivity | Specificity | NPV | |
| pTa | 89% | 86% | 99% |
| T1 | 97% | 86% | 99% |
| T2+ | 92% | 86% | 99% |
| G1 | 78% | 86% | 99% |
| G2 | 93% | 86% | 99% |
| G3 | 96% | 86% | 99% |
| NMIBC | 92% | 86% | 99% |
| MIBC | 92% | 86% | 99% |

GALEASTM Bladder
NMIBC
GALEASTM Bladder
MIBC
Cystoscopy (1,2)
Current Gold Standard


Sensitivity: 92%
Specificity: 86%
NPV: 99%
Sensitivity: 92%
Specificity: 86%
NPV: 99%
Sensitivity: 84%
Specificity: 86%
NPV: 96%
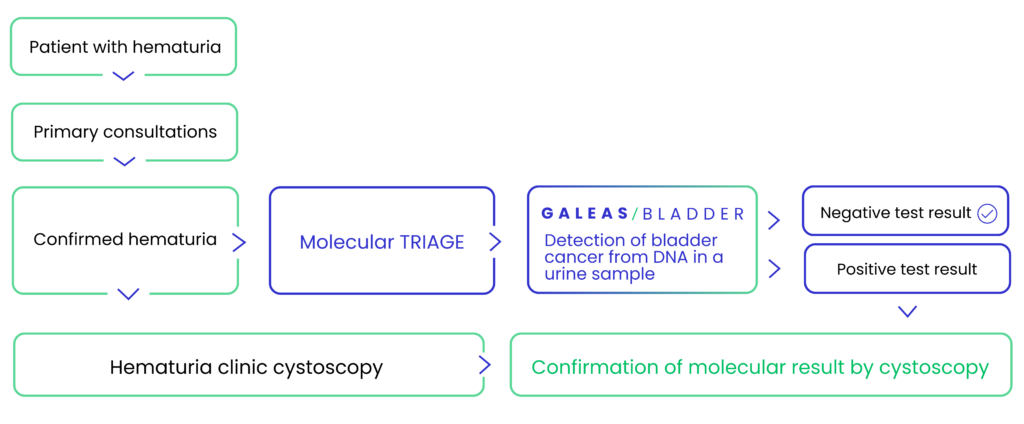
GALEAS™ Bladder for Hematuria Triage
Each year, hundreds of thousands of patients with hematuria (blood in urine) undergo invasive cystoscopy procedures, yet only about 10% are diagnosed with bladder cancer. GALEAS™ Bladder offers a non-invasive molecular triage solution, reducing unnecessary procedures and associated costs while providing rapid and accurate results.
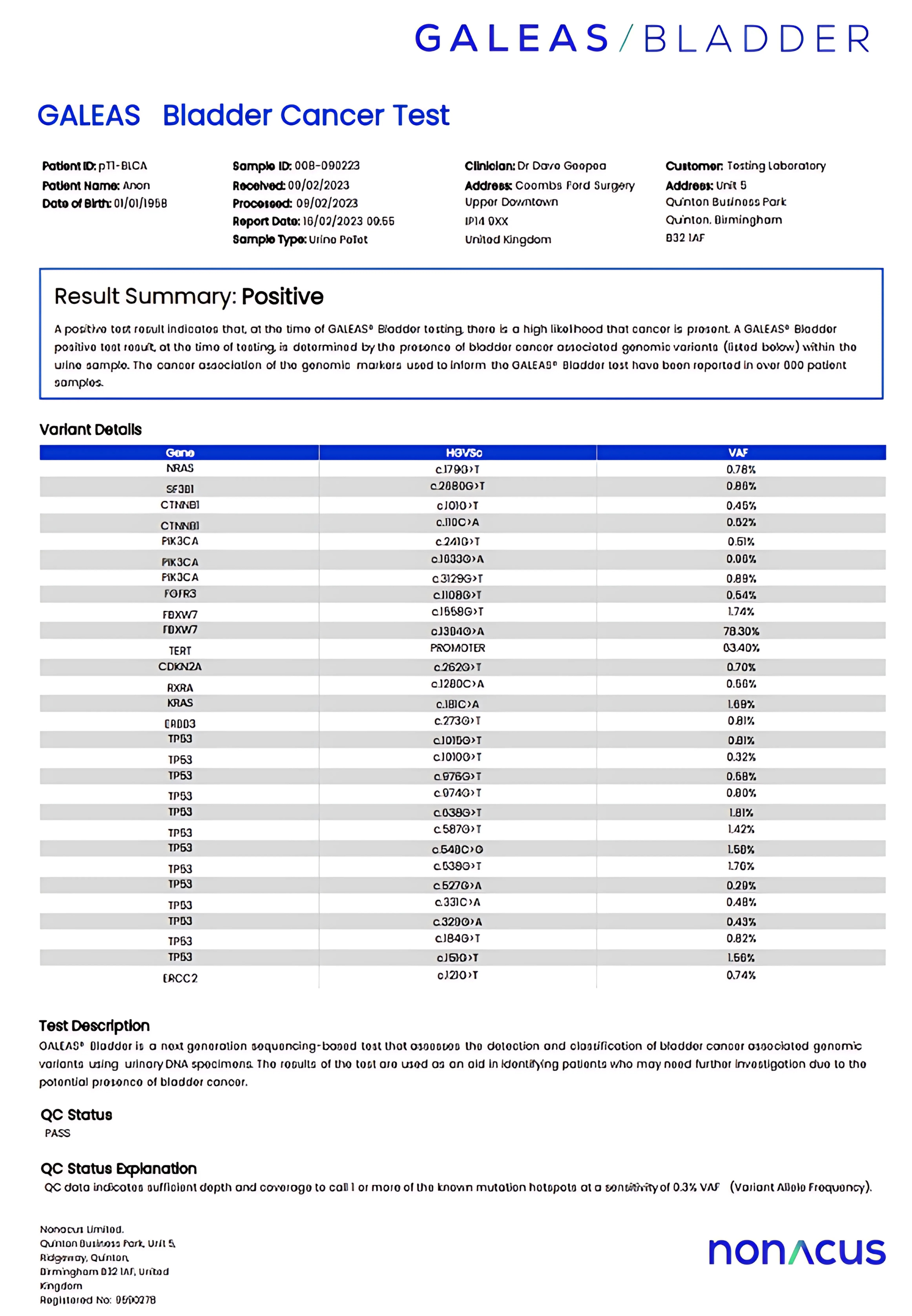
Reporting
- A positive/negative report will be sent to the urologist
- Details of somatic variants identified are provided
Cloud-Based GALEAS™ Analysis Software
The GALEAS™ platform provides:
- Simple positive/negative results
- Detailed somatic variant analysis
- Secure, cloud-based accessibility for seamless integration into clinical workflows



Urologist requests a urine collection device for patient
Patient sends barcoded urine sample to service laboratory
Lab uploads sequencing results to secure cloud-based GALEAS platform
Results analyzed, report generated and shared directly with urologist
Urologist shares results with patient and discusses next steps
References
1. Zhu, C. Z., Ting, H. N., Ng, K. H., & Ong, T. A. (2019). A review on the accuracy of bladder cancer detection methods. Journal of cancer, 10(17), 4038. Full article
2. Alimi, Q., Hascoet, J., Manunta, A., Kammerer‐Jacquet, S. F., Verhoest, G., Brochard, C., … & Peyronnet, B. (2018). Reliability of urinary cytology and cystoscopy for the screening and diagnosis of bladder cancer in patients with neurogenic bladder: a systematic review. Neurourology and Urodynamics, 37(3), 916-925. Full article
3. Ward et al. BJU Int. 2019, Targeted deep sequencing of urothelial bladder cancers and associated urinary DNA: a 23-gene panel with utility for non-invasive diagnosis and risk stratification
3. Ward DG, Gordon NS, Boucher RH, et al. Targeted deep sequencing of urothelial bladder cancers and associated urinary DNA: a 23-gene panel with utility for non-invasive diagnosis and risk stratification. BJU Int. 2019;124(3):532-544. doi:10.1111/BJU.14808. Full article
Ordering Information
| Ordering Information | Pack Size | Catalog Number |
|---|---|---|
| GALEAS™ urine collection device | 1 | NGS_GAL_UCD |
| GALEAS™ Bead Xtract: Urine gDNA extraction kit | 96 samples | PRE_GAL_BXG_96 |
| GALEAS™ Bladder kit (includes 1-96 indexes and complimentary analysis of FASTQ files using GALEAS™ analysis software for up to 96 samples) | 96 samples | NGS_GAL_BCP_FR_96_A |
| GALEAS™ Bladder kit (includes 97-192 indexes and complimentary analysis of FASTQ files using GALEAS analysis software for up to 96 samples) | 96 samples | NGS_GAL_BCP_FR_96_B |
| GALEAS™ Bladder kit (includes 193-288 indexes and complimentary analysis of FASTQ files using GALEAS analysis software for up to 96 samples) | 96 samples | NGS_GAL_BCP_FR_96_C |
| GALEAS™ Bladder kit (includes 289-384 indexes and complimentary analysis of FASTQ files using GALEAS™ analysis software for up to 96 samples) | 96 samples | NGS_GAL_BCP_FR_96_D |
| GALEAS™ Bladder validation control kit | 48 | NGS_GAL_GBVC_48 |